|
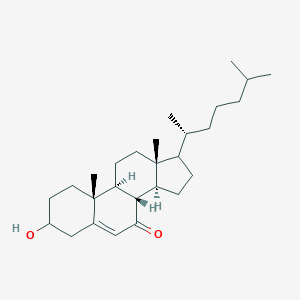
7-Ketocholesterol is a major oxidation product of cholesterol (oxysterol) found in human atherosclerotic plaque and is more atherogenic than cholesterol in some animal studies. Oxysterols (oxygenated forms of cholesterol) are present at low levels in the circulation and accumulate is plasma and tissues in some pathologies. In atherosclerotic lesions, 7-oxygenated oxysterols, predominantly 7-ketocholesterol, accumulate and have been implicated in the pathology of the disease. There is some in vivo and in vitro evidence that sterol 27-hydroxylase acts on 7-ketocholesterol to initiate its degradation to more polar, water-soluble products. Recent studies indicate an alternative mechanism, in which 7-ketocholesterol is reduced to 7 beta-hydroxycholesterol by 11 beta-hydroxysteroid dehydrogenase type 1. 7-Ketocholesterol can inhibit cholesterol 7 alpha-hydroxylase, the rate-limiting step in bile acid biosynthesis, as well as strongly inhibiting HMG-CoA reductase, the rate-limiting enzyme in cholesterol biosynthesis. It has even been suggested that 7-ketocholesterol is formed enzymically as an endogenous regulator of cholesterol biosynthesis. However, when tested as a pharmacological cholesterol-lowering agent, inhibition of HMG-CoA reductase was rapidly overcome and the 7-ketocholesterol metabolised. In vitro, 7-ketocholesterol has wide-ranging and potent effects, most of which have the potential to contribute to atherosclerosis. For example, 7-ketocholesterol can be cytotoxic and can induce apoptosis in vascular cells. These effects, either individually or more likely, in combination, all implicate 7-ketocholesterol in the initiation and development of atherosclerosis, but further work is needed to establish whether or not its role is a direct causal one. 7-Ketocholesterol is the second most abundant oxysterol found in human atherosclerotic plaque, after the enzymically formed 27-hydroxycholesterol (cholest-5-ene-3beta,27-diol). 7-Ketocholesterol differs from cholesterol by a ketone functional group present at the 7-position. It is produced from cholesterol via the epimeric cholesterol 7-hydroperoxides (cholest-5-ene-3beta-ol-7-hydroperoxide) which decompose to the epimeric 7-hydroxycholesterols (cholest-5-ene-3beta,7-diol) and 7-ketocholesterol. 7-Ketocholesterol is a major dietary oxysterol. It has also been widely suggested that 7-ketocholesterol present in atherosclerotic tissue may be derived from the diet. Certainly, 7-ketocholesterol is a major oxysterol found in cholesterol-rich processed foodstuffs. Dietary 7-ketocholesterol is rapidly metabolised by the liver to 7beta-hydroxycholesterol (cholest-5-ene-3beta,7beta-diol), unusual bile acids and perhaps even cholesterol itself. Its conversion to 7beta-hydroxycholesterol is well documented.
|
 click to show the details of this protein
click to show the details of this protein
 click to show the details of experiment for validating this pair
click to show the details of experiment for validating this pair